Proper Water Sampling Practise Prior to Metals Determination
Published: May 2023
Download PDF
EnviroMail_03_Europe_Proper Water Sampling Practise Prior to Metals Determination
ALS helps maximize safety, data quality and efficiency
Sampling for trace metals determination in groundwaters, surface waters, leachates or in process
and waste waters is a crucial moment of metals analysis. This EnviroMail will provide background
information and technical guidance on trace metals preservation and sampling intended for total
and dissolved metals determination.
Sampling containers and their preservation
One of two most important aspects of trace metals analysis is the quality of the sample container, which ensures maximum integrity of the data obtained in the laboratory. The sample containers for metal analysis used by ALS are made of high quality plastic (HDPE: High Density PolyEthylene). The material used to produce these samplers has been shown not to affect the analysis of trace metals. Containers designated for metals analysis are pre-filled/fixed with nitric acid and are delivered by ALS laboratories to clients for sampling.
The nitric acid preservative solution is added to the sample tubes to ensure sample stability, where its presence will prevent inadvertent precipitation of metals, adsorption of metals to the samplers’ walls, and/or any other colloidal material or sediment that is part of the sample being collected. Fixation thus helps to maintain the original metal concentration in the sample during its transport to the laboratory.
Field filtration of water samples is the best way
The analysis of trace metals in water samples can be divided into basic analysis of total trace metals or analysis of dissolved metals. Dissolved metals in water exist in complex equilibria that are easily influenced by many physicochemical factors such as pH, temperature, or ekvilibrium redox conditions. Even a small change in these conditions can cause precipitation, co-precipitation of metals, sorption, or dissolution of particles. The resulting metal concentrations may thus obtain a significant positive or negative deviation from the correct results.

Instantaneous filtration of water samples at the point of collection (field filtration) has been shown to
significantly stabilise the behaviour of metals. Thus filtration helps to avoid an undesirable reactions, by which the elements iron, arsenic, lead, cadmium, and the total dissolved metals can be highly affected. In some cases, losses can be significant. On the other hand, field filtration also prevents unwanted release of metals from the clogged sediment, which could significantly increase the concentration of metals present.
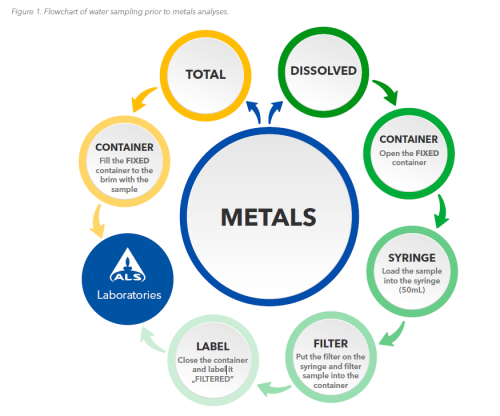
Ideal sampling procedure:
ALS recommends and supplies the same sampling containers for both total and dissolved metals
determination. Determination of total metals Sampling water directly into the fixed (nitric acid) 60mL
HDPE container, sample filtration is NOT performed for this type of analysis.
Determination of dissolved metals
The recommended procedure is to filter the samples collected in the field directly into the fixed 60mL HDPE containers. The container should be marked or labelled to indicate that the sample has already been filtered.
Note: for dissolved metals analysis, there is also the option of collecting the unfiltered sample in an unfixed container. Filtration and fixation is then carried out in the laboratory, but the results may be affected as described above.
-
Only good practise of sampling procedure can avoid false positive or negative results.
-
The use of fixed ALS containers and field filtration of water samples is of utmost importance
Advantages of use small 60ml-HDPE fixed- containers:
- Usually only one filter and syringe set is needed to filter 50-60 mL of water, so the sample is homogeneously processed and the risk of false positive or negative results is reduced.
- The smaller amount of water that is mixed with the nitric acid contained in the sample tube does not create a risk of exothermic reactions due to the smaller amount of diluted acid.
- Faster sampling process in the field, improving efficiency and reducing the time required for sample collection and filtration.
- Small 60mL vials are particularly advantageous for low-flow sampling, saving sampling time.